Atebimetinib (IMM-1-104) + Modified Gemcitabine/Nab-Paclitaxel (mGnP) Shows Extraordinary Overall Survival (OS) and Favorable Tolerability in First-Line (1L) Metastatic PDAC: Updated Phase 2 Results
Deep Cyclic Inhibition of MEK: A Transformational Approach to Durable and Safe Combinations in Ras-Mutant Cancers
Potential anti-cachexia properties of novel dual-MEK inhibitor IMM-1-104
Phase 1 Interim Population PK/PD Modeling and Recommended Phase 2 Dose Exploration for IMM-1-104, A Novel Concept Oral Deep Cyclic Inhibitor of MEK
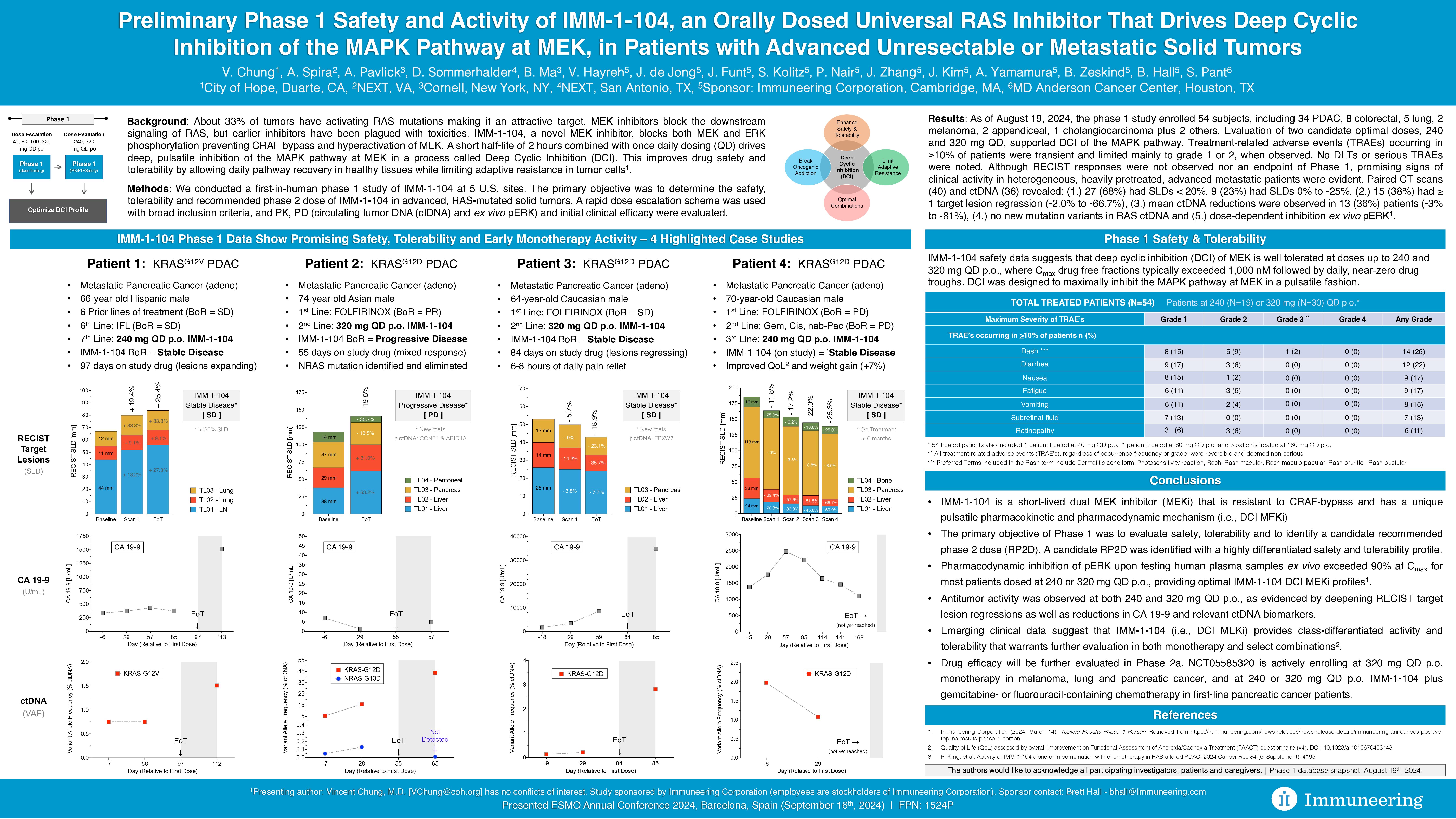
Preliminary phase 1 safety and activity of IMM-1-104, an orally dosed universal RAS inhibitor that drives deep cyclic inhibition of the MAPK pathway at MEK, in patients with advanced unresectable or metastatic solid tumors
Vincent Chung, Alex Spira, Anna Pavlick, David Sommerhalder, Brett Hall, Vinny Hayreh, Jan de Jong, Jason Funt, Sarah Kolitz, Praveen Nair, Peter King, Jenny Zhang, Jason Kim, Amy Yamamura, Benjamin Zeskind, Shubham Pant
Background:
About 33% of tumors have activating RAS mutations making it an attractive target. MEK inhibitors block the downstream signaling of RAS, but earlier inhibitors have been plagued with toxicities. IMM-1-104, a novel MEK inhibitor, blocks both MEK and ERK phosphorylation preventing CRAF bypass and hyperactivation of MEK. A short half-life of 2 hours combined with once daily dosing (QD) drives pulsatile inhibition of the MAPK pathway at MEK in a process called Deep Cyclic Inhibition (DCI). This improves drug safety and tolerability by allowing daily pathway recovery in healthy tissues while limiting adaptive resistance in tumor cells.
Methods:
We conducted a first-in-human phase 1 study of IMM-1-104 at 5 U.S. sites. The primary objective was to determine the safety, tolerability and recommended phase 2 dose (RP2D) of IMM-1-104 in advanced, RAS-mutated solid tumors. A rapid dose escalation scheme was used with broad inclusion criteria, and PK, PD, ctDNA and initial clinical efficacy were evaluated.
Results:
As of May 1, 2024, the phase 1 study completed enrollment of 45 subjects, including 30 PDAC, 5 colorectal, 5 lung, 2 melanoma, 1 cholangiocarcinoma plus 2 others. Two candidate optimal doses, 240 and 320 mg QD, supported ≥ 90% DCI of the MAPK pathway at ERK. Treatment-related adverse events (TRAEs) occurring in ≥10% of patients were mild and transient (see table). No dose-limiting toxicities or serious TRAEs were observed. Early signs of clinical activity in a heavily pretreated, advanced metastatic patient population were promising and paired CT scans (32) and ctDNA (27) revealed: (1.) 21 (66%) RECIST SLD < 20%, 8 (25%) with SLD’s ≤ 0%, (2.) 13 (41%) had ≥ 1 target lesion regression (-4.8% to -50.0%), (3.) reductions in mean VAF ctDNA observed in 9 (33%) patients (-24% to -81%), and (4.) no new variants in RAS ctDNA emerged.
Conclusions:
Disrupting MAPK-pathway addicted tumors at MEK with IMM-1-104 using a novel DCI approach was well tolerated and demonstrated lesion and molecular level responses as monotherapy in an advanced, heavily pretreated phase 1 patient population. Phase 2a mono and combo arms is underway.
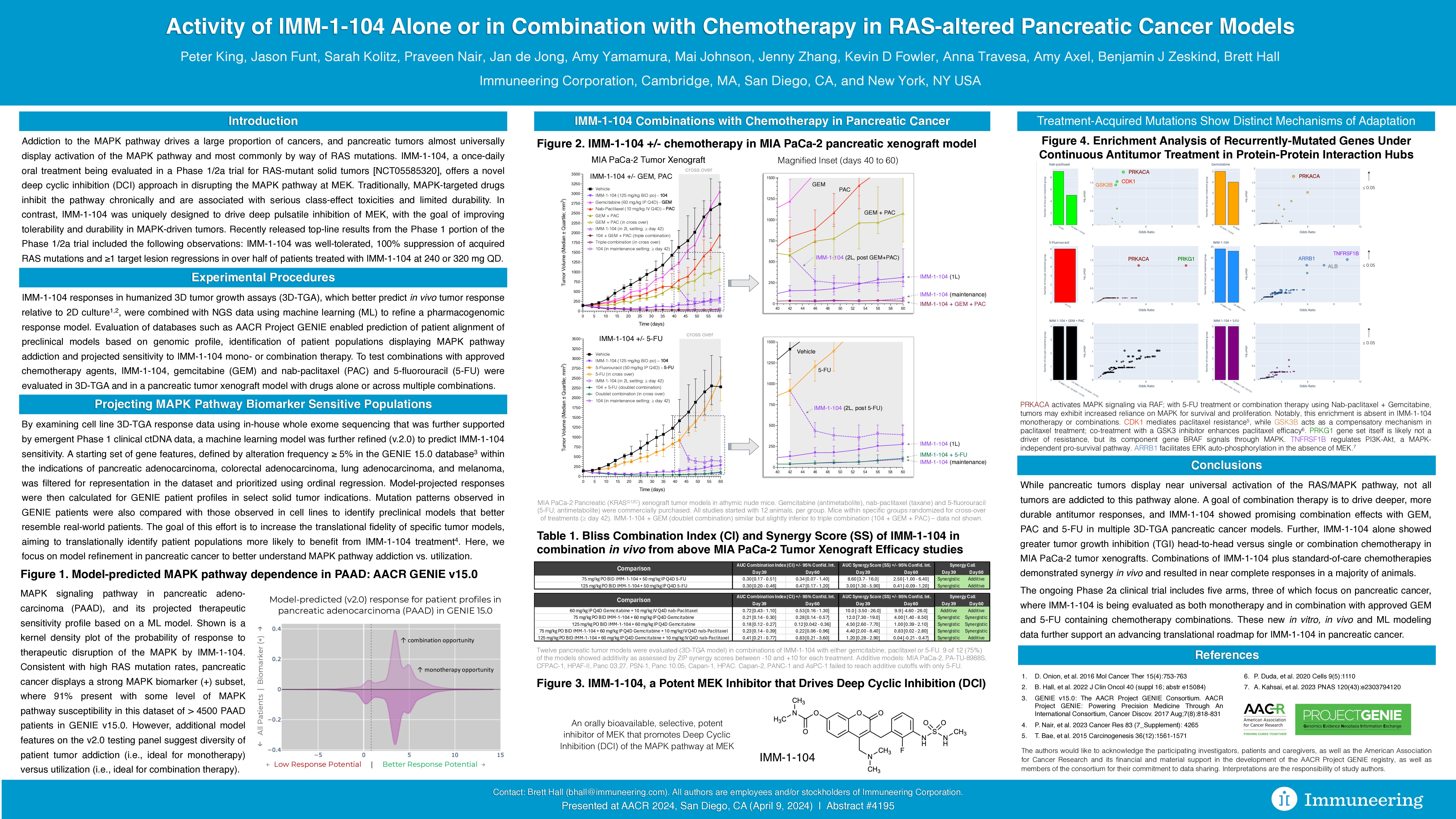
Activity of IMM-1-104 alone or in combination with chemotherapy in RAS-altered pancreatic cancer models
In a poster titled, “Activity of IMM-1-104 alone or in combination with chemotherapy in RAS-altered pancreatic cancer models,” IMM-1-104, gemcitabine (GEM), nab-paclitaxel (PAC), and 5-fluorouracil (5-FU) were evaluated in tumor xenograft models alone or across multiple combinations.
Results:
- IMM-1-104 showed promising combination effects when treated with gemcitabine (GEM), paclitaxel (PAC) or fluorouracil (5FU) in 3D-tumor growth assay (TGA) pancreatic cancer models.
- IMM-1-104 was synergistic with chemotherapy in animal models of pancreatic cancer.
- In a human pancreatic cancer cell line (MIA PaCa-2) tumor xenograft model, IMM-1-104 alone showed greater tumor growth inhibition (TGI) and better durability than any single or combination chemotherapy tested.
- At day 39, antitumor activity (TGI%) was 103% for IMM-1-104 at 125 mg/kg BID PO, 25.2% for GEM at 60 mg/kg IP Q4D, 62.2% for PAC at 10 mg/kg IV Q4D, and 36.6% for 5FU at 50 mg/kg IP Q4D.
In the Phase 2a portion of Immuneering’s ongoing IMM-1-104 Phase 1/2a clinical trial, IMM-1-104 is being evaluated as both monotherapy and in select combinations with approved chemotherapeutic agents. The Phase 2a portion includes five arms, three of which focus on patients with pancreatic cancer. Patients are now on treatment in multiple arms of the ongoing Phase 2a trial, including multiple patients with pancreatic cancer who are being treated with IMM-1-104 in combination with chemotherapy in the first-line setting. The company expects initial data from multiple Phase 2a arms in 2024.
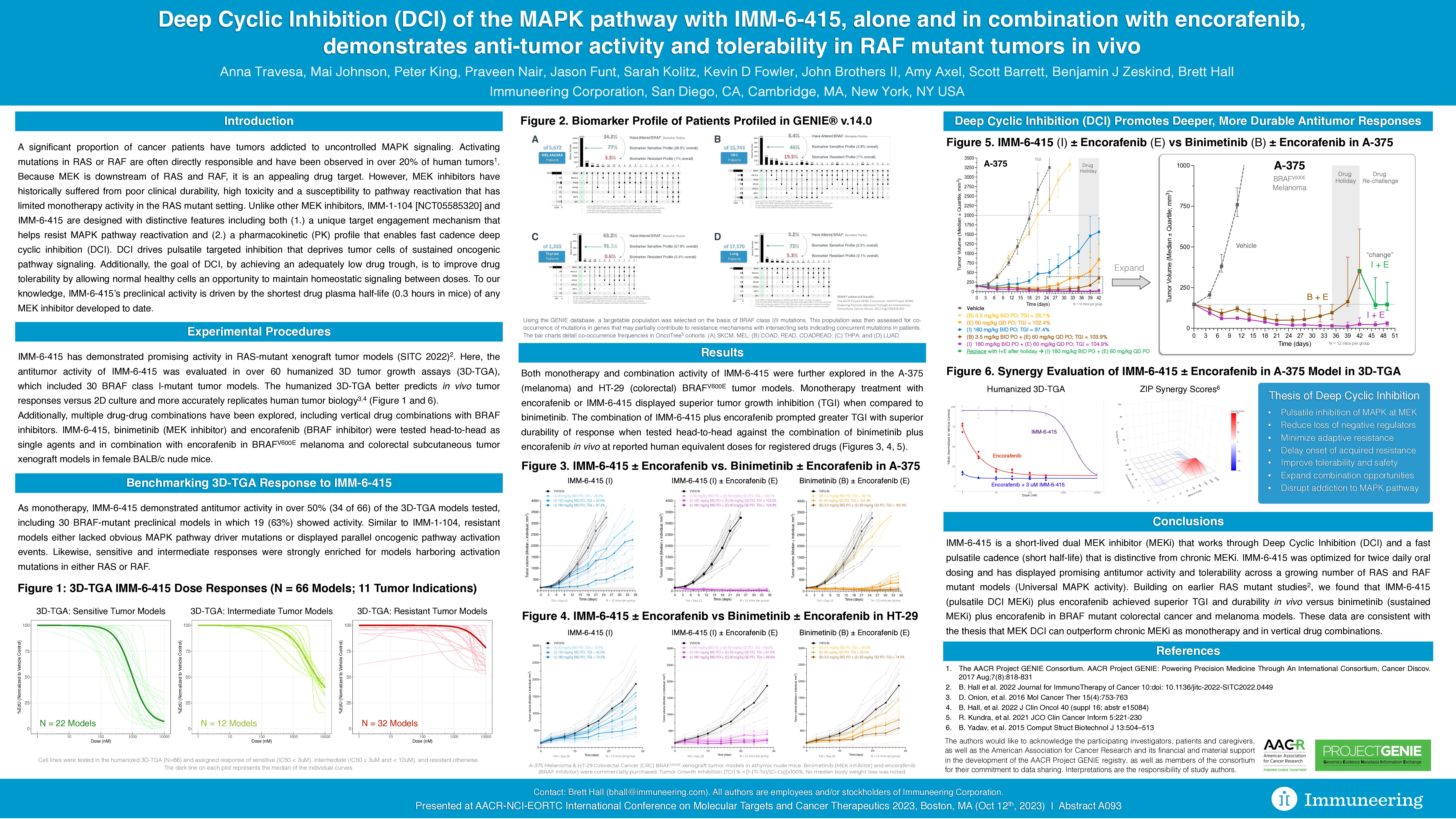
Deep Cyclic Inhibition of the MAPK pathway with IMM-6-415, alone and in combination with encorafenib, demonstrates anti-tumor activity and tolerability in RAF mutant tumors in vivo
- Anti–tumor activity of IMM-6-415 was evaluated in more than 60 humanized 3D-TGA models, which included 30 BRAF class I-mutant tumor models. Multiple drug-drug combinations have been explored, including vertical drug combinations with BRAF inhibitors.
- IMM-6-415, binimetinib and encorafenib were tested head-to-head as single agents and in combination with encorafenib in BRAFV600E melanoma and colorectal subcutaneous tumor xenograft models in female BALB/c nude mice.
- As monotherapy, IMM-6-415 demonstrated anti–tumor activity in over 50% (34 of 66) of the 3D-TGA models tested, including 30 BRAF mutant preclinical models in which 19 (63%) showed activity. Similar to IMM-1-104, resistant models either lacked an obvious MAPK pathway driver mutation or displayed parallel oncogenic activation events. Sensitive and intermediate responses were also strongly enriched for models harboring an activation mutation in RAS or RAF.
- Monotherapy treatment with encorafenib or IMM-6-415 displayed superior TGI when compared to binimetinib in the A-375 (melanoma) and HT-29 (colorectal) BRAFV600E tumor models.
- In combination with encorafenib, IMM–6–415 achieved a greater TGI in vivo than the combination of encorafenib plus binimetinib in BRAFV600E colorectal cancer and melanoma tumor models, suggesting an opportunity for IMM-6-415 as monotherapy or in combination regimens for the treatment of BRAF mutant tumors.
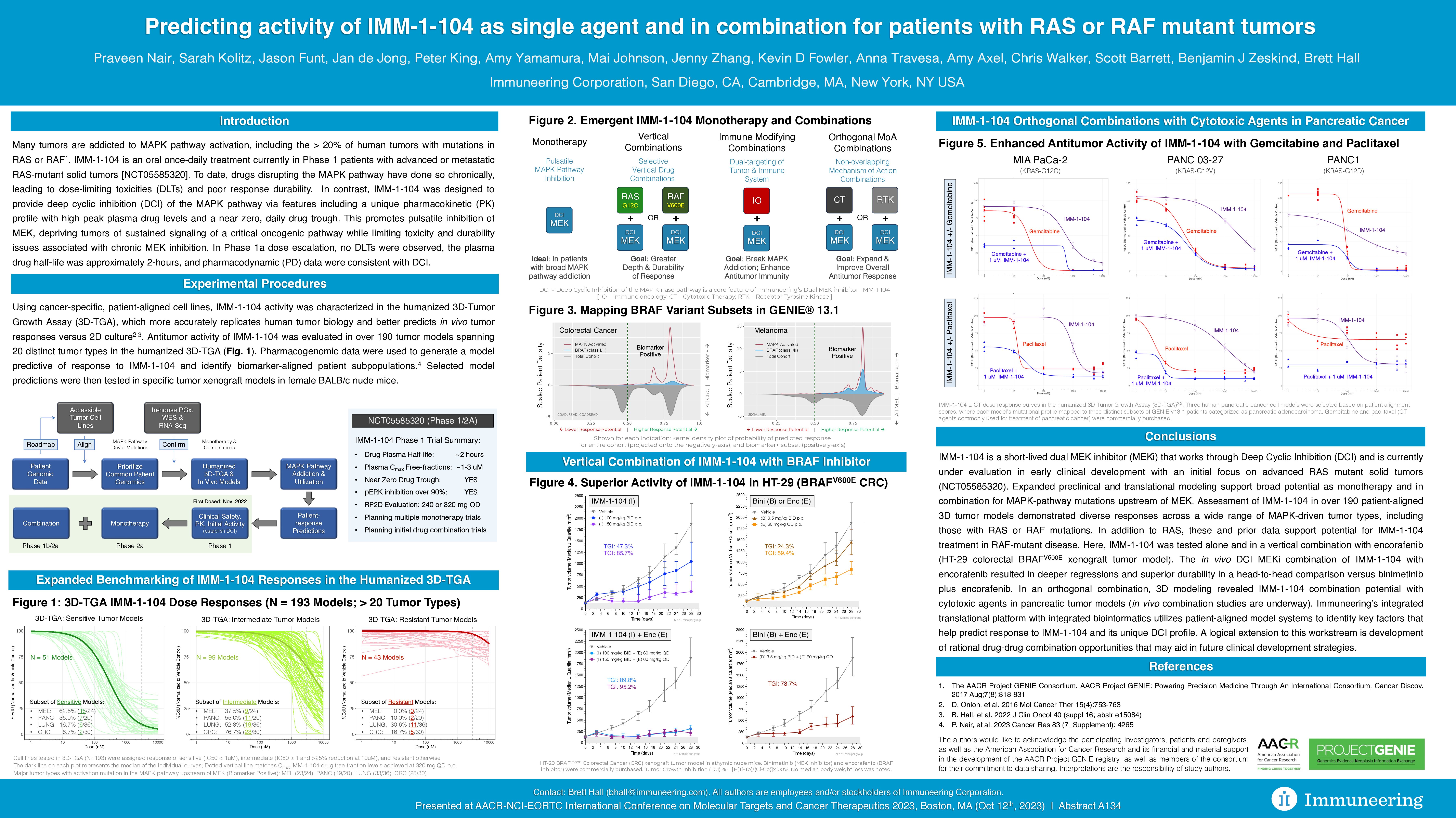
Predicting activity of IMM-1-104 as single agent and in combination for patients with RAS or RAF mutant tumors
- Anti-tumor activity of IMM-1-104 was characterized in 193 tumor models spanning 20 distinct tumor types in the humanized 3D-tumor growth assay (3D-TGA) using cancer-specific, patient-aligned cell lines.
- IMM-1-104 demonstrated diverse responses across a wide range of MAPK-driven tumor types, including those with RAS or RAF mutations.
- Pharmacogenomic data were used to generate a model predictive of response to IMM-1-104 and identify biomarker-aligned patient subpopulations.
- Sensitivity to IMM-1-104 (IC50 < 1uM) tested in 3D-TGA cell lines was highest in melanoma (62.5%) followed by pancreatic cancer (35.0%) and lung cancer (16.7%).
- IMM-1-104 was tested in combination with gemcitabine or paclitaxel in humanized 3D models of pancreatic cancer, demonstrating enhanced activity and combination therapy potential.
- IMM-1-104 in combination with encorafenib drove deeper regressions and superior durability of response in a head-to-head in vivo comparison versus binimetinib plus encorafenib. Tumor growth inhibition (TGI) was between 89.8% and 95.2% with the IMM-1-104 plus encorafenib combination and 73.7% with the binimetinib plus encorafenib combination.
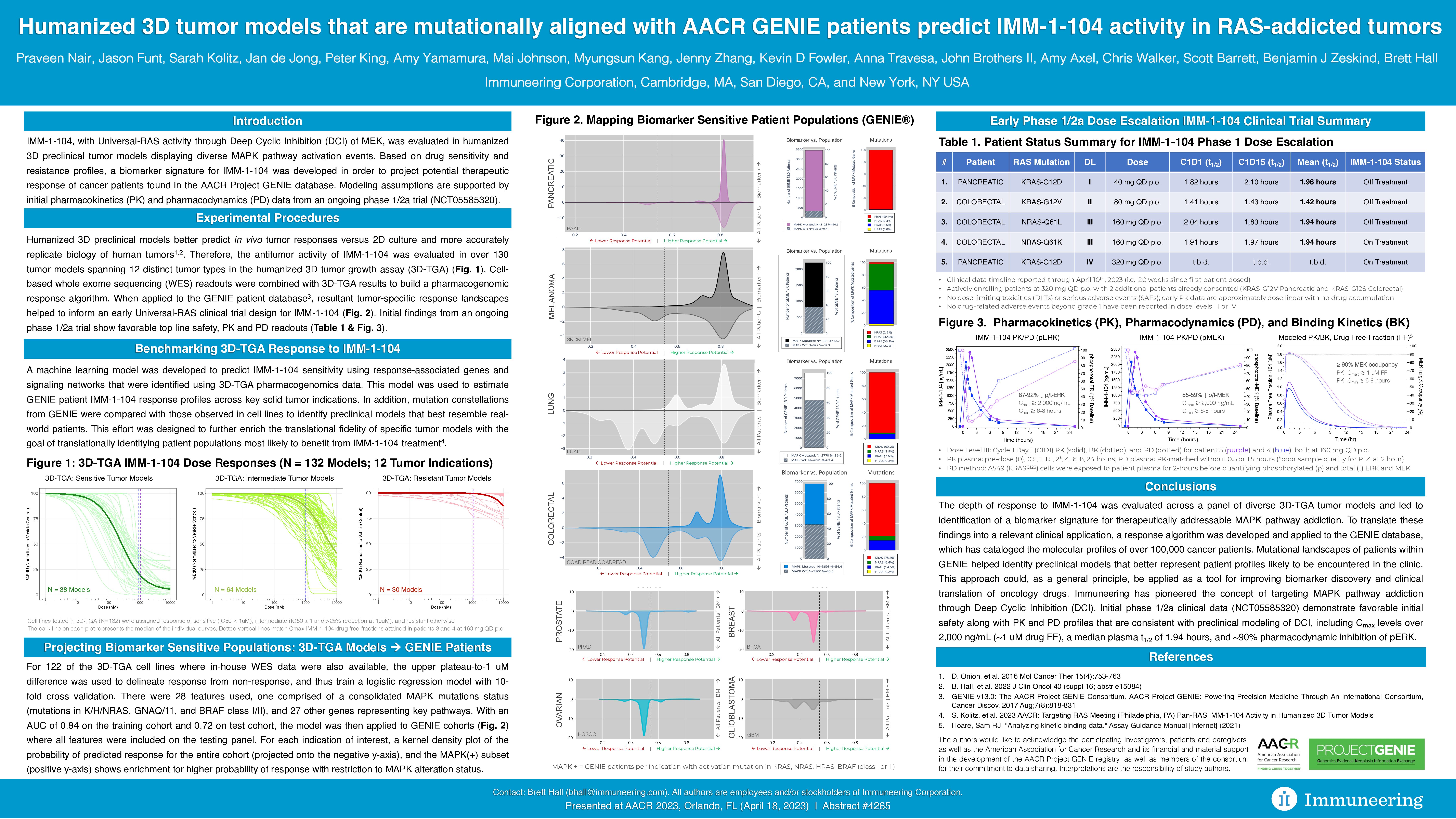
Humanized 3D tumor models that are mutually aligned with AACR GENIE patients predict IMM-1-104 activity in RAS-addicted tumors
Highlights of the initial IMM-1-104 Phase 1 PK, PD and safety data presented at AACR include (as of data cut-off date of April 10, 2023, including patients with pancreatic and colon cancer):
- Significant PK Cmax levels (plasma concentration of therapy in a specific area of the body) observed with IMM-1-104 of over 2,000 ng/mL (or approximately 1 uM drug free-fraction at 160 mg once daily oral dose)
- Greater than 90 percent PD inhibition of phosphorylated extracellular signal-regulated kinase (pERK) with IMM-1-104 compared to pretreatment baseline for patients at the third dose level (160 mg once daily oral)
- A median plasma half-life (t1/2) of 1.94 hours observed with IMM-1-104 across the first three dose levels evaluable (40 mg, 80 mg and 160 mg once daily oral), in patients with pancreatic and colorectal cancer with different RAS mutations, including KRAS-G12D, the most common mutation present in pancreatic cancer
- IMM-1-104 was well tolerated with no DLTs or SAEs observed and no drug-related adverse events beyond Grade 1 observed
Title: Humanized 3D tumor models that are mutually aligned with AACR GENIE patients predict IMM-1-104 activity in RAS-addicted tumors
Date: Tuesday, April 18, 2023, 9:00am – 12:30pm ET
Poster session: AACR Project GENIE Use Cases
Abstract Number: 4265